Apple’s ResearchKit Takes Medical Research Years Into the Future
Apple just released what history might say is its most important product ever.
And guess what? Hardly anyone even knows it exists.
It’s called ResearchKit. It’s a software toolkit that lets researchers write iPhone apps for medical studies.
Yawn, right?
No. This is a huge deal.
Every year, people buy 1 billion smartphones and 70 million wearable health trackers. These devices monitor how much we move and sleep, and where we are in the world. Some of them track our pulse, stress, posture, sunlight exposure, perspiration, and so on.
Yet how much of this data is available to scientists? None of it. These phones and trackers spew out terabytes of useful data a day — and scientists can’t get at it, analyze it, or parse it to draw conclusions about medicine and health. We’re conducting the biggest clinical trial in human history, and we are throwing away the results.
How studies are conducted now
What is science, anyway? It’s “the systematic study of the structure and behavior of the physical and natural world through observation and experiment.”
Without observation and experiment, science doesn’t move forward. That’s why medical studies are so important. No medical breakthrough ever came about without one.
Today, studies on disorders, medicines, and treatments are conducted the same way they have been for 50 years. To get research subjects, a researcher seeks participants by putting up fliers or taking out an ad in a newspaper. It’s low-tech and absurdly inefficient.
Probably the most quoted line from Apple’s ResearchKit promotional video is this one, from Kathryn Schmitz, PhD, of the University of Pennsylvania Health System: “We have sent out over 60,000 letters. Those 60,000 letters have netted 305 women.”
Once you’ve found your subjects, you sit them down and go over the risks and benefits — a paperwork nightmare that takes at least half an hour per participant. Right there, you’ve limited the scope of your study, and limited it to people who live nearby.
That’s the world of medical research right now. Slow, small-scale, inefficient.
Consent and privacy
Apple worked with five medical institutions, associated with places like Harvard, Stanford, and Mount Sinai in New York City, to come up with the first five ResearchKit-based apps. They represent studies of asthma, diabetes, heart disease, Parkinson’s, and breast cancer. (You can enroll in the heart, breast cancer, and Parkinson’s studies even if you don’t have those diseases. The researchers will use your data as a control.)
The first time you open a ResearchKit app, you encounter the first huge breakthrough: The sign-in process is electronic. No more researcher sitting across from a table asking you questions and going over paperwork.
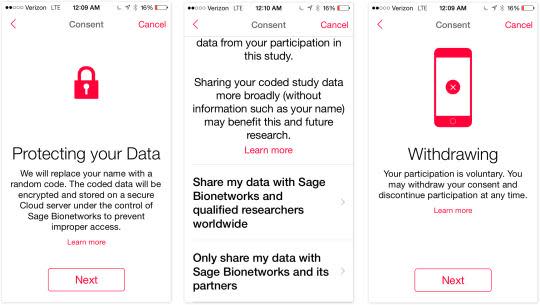
Ordinarily, that sort of enrollment process would never fly with the hospitals’ notoriously conservative IRBs — the institutional review boards whose job it is to approve every step of a medical study, making sure it complies with all medical, ethical, and legal concerns.
But by choosing five institutions to help launch ResearchKit, Apple wound up creating competitive pressure. “No institution wanted to be left out because its review board wouldn’t approve it,” says Dr. Eric Schadt of Mount Sinai, whose team created the Asthma Health app. The result: All five institutions’ IRBs approved an electronic consent-granting process within the app. With just this one advancement, the world of medical research has changed forever.
In any case: After you read the consent forms, you’re told exactly who will get to see your data and for what purpose it will be used. You can opt out of any part of the data collection at any time. Indeed, you can drop out of a study at any time, although you agree to let the researchers use the data you’ve generated up to that point.
In each app, you’re also asked: Do you grant permission for us to share your data with medical researchers at other institutions, conducting other studies?
Incredibly, Schadt says that 75 percent of the people who’ve signed up for his asthma study have agreed to let their data be shared more widely. They truly want to help.
So, no question: The consent and sign-up process is complete and thorough and patient. If you ask me, it’s too much so. You’re shown the details of the study, your privacy, and your freedoms, one screen at a time. Then you take a quiz to confirm that you get it. Then you get an email saying the same thing for a third time. We get it!
By the way: Apple says that Apple never gets your data. I asked Dr. Schadt if that’s true.
“Apple never touches the data,” he confirmed. “The data’s collected in the app, and the data goes to what we call a bridge server. It’s maintained by Sage Bionetworks, a nonprofit medical research institute in Seattle. It’s all encrypted and very secure, meets all the industry standards for shipping sensitive data, HIPAA compliant. From the bridge server, it goes into the server we access. The only people who have access to the data are the investigators of the study.”
Size and scope
By now, you realize why ResearchKit is such a big deal: Because it hugely amplifies the size and scope of a study. Instead of generating some data, it generates big data. You can sign up a lot of people for your study, fast.
Using traditional recruitment techniques, “you’re lucky if you get 300 or 500 over the course of a year,” Schadt told me. “If you’re super lucky, over many years, you’ll get a thousand enrolled in a study — and they’ll be geographically limited.”
But he got 7,500 asthma sufferers enrolled in his study within the first month. “That’s astonishing — it just blew us away.”
Furthermore, the data they generate is continuous. In traditional studies, Schadt says, “You’re lucky if those individuals come in once a year, for a 30-minute visit. But with the app, we can collect that data continuously over time, providing a much deeper characterization of what’s going on.”
The other first-wave ResearchKit researchers told me similar stories. For example, the breast-cancer app, Share the Journey, had 6,000 downloads and 2,500 enrollments in the study within three weeks.
For the Stanford heart study, a mind-blowing 30,000 people enrolled through the MyHeart Counts app in the first month of availability.
And even that is just the beginning, says Dr. Mike McConnell, whose team created the app. “We exceeded the 10,000 threshold the first 24 hours, but with the number of iPhone users around the world, we’d really like to reach many more people. There is clearly a process of how to make them as user-friendly and engaging as possible, to encourage and maintain engagement.”
2-way studies
I visited Schadt and his team at Mount Sinai in New York, to get a better idea of how his app works — and what the data looks like when it comes to him.

The app itself is very simple. Each day, it prompts you to answer three or four questions about how you feel, if you used your inhaler today, and if you took your medication. Behind the scenes, it’s also recording your GPS location. From that, the app can figure out the local pollution levels you are exposed to: pollen count, ozone, heat index, and other asthma-triggering conditions.
This is typical for a ResearchKit app. The patient supplies some of the data by answering questions. The rest is generated automatically from the phone’s sensors: microphone, camera, accelerometer, GPS, gyroscope, and so on. If you have an external health device that talks to Apple’s HealthKit protocol — a fitness-tracker band, digital thermometer, Bluetooth scale, Bluetooth blood-pressure cuff, Bluetooth inhaler, and so on — that data can become part of the ResearchKit app’s data, too.
(Schadt notes that Bluetooth inhalers are already available. As they become popular and HealthKit-compatible, more of his app’s data can become auto-generated — and more reliable than the patient’s memory.)

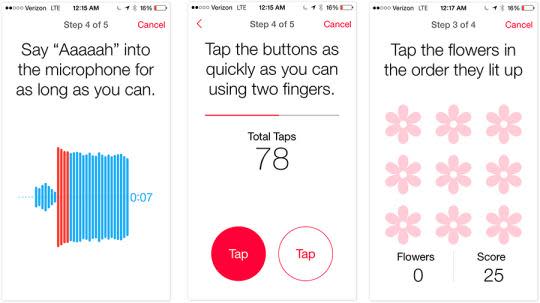
The mPower app for Parkinson’s is a great example. Each day, it asks you to participate in certain activities. One asks you to say “ahhh” into the microphone for as long as you can on a single breath. One asks you to tap the screen alternately with two fingers as fast as you can. One tracks your short-term memory by testing to see if you can remember a pattern of flowers lighting up.
Once the researchers start collecting data from their apps, they can slice and dice the results in astonishing ways. At Mount Sinai, for example, Schadt showed me a very visual graphic dashboard program on his laptop that displays all of the data from his 7,500 participants. With a click, he can view subsets of data — by state, age, ethnicity, smoking status, asthma medication, and so on.
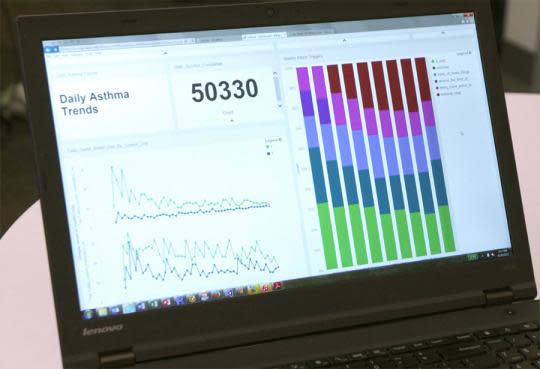
(One example I loved: His dashboard includes an aggregated graph that shows what events have triggered the participants’ asthma attacks each week. In states like California and Texas, common triggers include heat, exercise, and feathery animals. But in New York and New Jersey — and only those states — a frequent trigger turns out to be anger.)
We already know that pollution and pollen can trigger asthma attacks, of course. But we know only that those triggers affect the population of asthma sufferers. “What may be very severe for one person may be mild for another,” Schadt says. “We know it on a population level, but we want to get to a precision-based medicine. If we could start stratifying the patient population into different groups, we would then monitor them differently, inform them differently, and manage their care differently.”
In other words, these apps could hurry us along into the next generation of medical care, where your treatment is tailored to your DNA, geography, and bodily responses — instead of blindly trying one drug, then another, in hopes of eventually finding something that works.
All of the researchers I interviewed said the same thing: that a huge advantage of using an app to conduct the study is that it can become two-way. In a regular study, you provide data to the researchers — and then you never hear from them again.
But ResearchKit apps can begin to give you advice based on your medication situation.
For example, McConnell’s app, MyHeart Counts, is part of a study that examines relationships between activity, motivation, risk factors, and heart health. The app tracks your activity, either from the phone’s motion sensors or HealthKit-compatible wearables, for a week. You also take a six-minute walking test four times a year.
After three months, the app starts giving you personalized motivational prompts to get you to move more.
Apple’s motivation
This is great and all — but why is Apple doing it?
Your first assumption might be: “to sell iPhones, of course.”
But here’s the thing: Apple has made ResearchKit open source. Apple invites anyone to use it or even modify it — even Apple’s rivals, like Samsung, Google, and Microsoft.
In fact, Apple assisted with the writing of those first five apps on the conditionthat the apps themselves also be open-source. So they are.
This is a gigantic move. It means that Apple gets nothing out of this — except the rosy glow of doing something good for humanity, and maybe a little good PR.
Room for improvement
All of the researchers are positively giddy about ResearchKit’s potential. “Sage, Stanford, Harvard — we all talk pretty regularly,” says Schadt, referring to the institutions that made the first five ResearchKit apps. “Everybody is just over-the-top blown away at the response. Everybody’s going, ‘Wow, this is really changing the mindset of research.’”
That doesn’t mean there aren’t critiques, however. One example jumps out pretty quickly: These studies won’t include any data from patients who can’t afford a smartphone.
Some of the apps, like Asthma Health and GlucoSuccess (for diabetes), require that you actually have that disease to participate. But the app has no way to confirm that you’re not just a merry prankster, skewing the data by faking it.
ResearchKit apps currently can’t incorporate genetic information, either, and they don’t integrate with patients’ electronic medical records. Furthermore, plenty of external medical sensors still don’t work with HealthKit and so can’t generate data for ResearchKit.
And, of course, ResearchKit apps can’t do things like scan your body or analyze fluid samples. Maybe in version 2.0.
The ResearchKit future
Still, taking medical research digital with ResearchKit promises to be just as disruptive a change as when music went digital. We can answer questions now thousands of times faster, with much bigger test samples, than we could in the posting-fliers age.
Stanford’s McConnell sums it up well: “ResearchKit solves a number of the current challenges to clinical research. How do you reach/recruit a large number of people? How to do help bring down the costs of large clinical research studies? How do you reach a broad geographic population? How do you make it easier and more engaging for people to participate? How can you collect more continuous, real-world, quantitative data? How can you give participants more feedback about their data, and the choice to share their research data broadly?”
Dr. Patricia Ganz, whose Share the Journey app is part of a UCLA breast-cancer study, agrees. “This is a proof of principle,” she told me. “It’s a new way to conduct research. It’s also amazing that Apple has donated the resources to make it happen.”
Mount Sinai’s Schadt already has 10 more apps in development for other diseases. I’ll give him the last word: “You’re not only going to maintain a much healthier, more productive person, but you’ll save the health system a lot of money as well. That’s where I think this is all going to have a really dramatic impact on public health.”

