Health Canada licensing of natural remedies 'a joke,' doctor says
Popular over-the-counter remedies, approved by Health Canada as "safe and effective," may be supported by little to no scientific evidence that the products work, an investigation by CBC’s Marketplace reveals.
Canadians spend $2.4 billion a year on natural health products.While some products may have clinical trials or other scientific evidence to support their claims, many do not require any scientific proof, and there’s little way for consumers to tell the difference.
"It's frustrating that the government standards are not protecting the public the way they should be," Dr. Matthew Stanbrook, deputy editor of the Canadian Medical Association Journal (CMAJ), told Marketplace co-host Erica Johnson.
This is "lending the veneer of approval to something that really hasn't demonstrated the science."
To test how little evidence is required to get Health Canada to license a product, Marketplace created a children’s remedy, applied for approval and received a licence.
Manufacturers need a Health Canada licence number in order to sell their natural health product. Once a product is licensed, manufacturers can claim it is approved as "safe and effective."
The investigation found that even a remedy making serious health claims — such as the ability to reduce children’s fever — can acquire a licence with no scientific evidence.
Process is 'a joke'
Pharmaceutical drugs require years of clinical research before they are approved for sale. But Health Canada allows natural health product manufacturers to make similar health claims based on traditional medicine or homeopathic use, instead of scientific evidence.
Licensed natural products have an eight-digit Natural Product Number (NPN) or Homeopathic Medicine Number (DIN-HM) on the label, which may give consumers peace of mind knowing the product has been authorized and approved for sale.
Last year, Health Canada licensed almost 10,000 natural health products, including herbal remedies, traditional medicines and homeopathic products. Many natural products are sold in drugstores alongside pharmaceutical options.
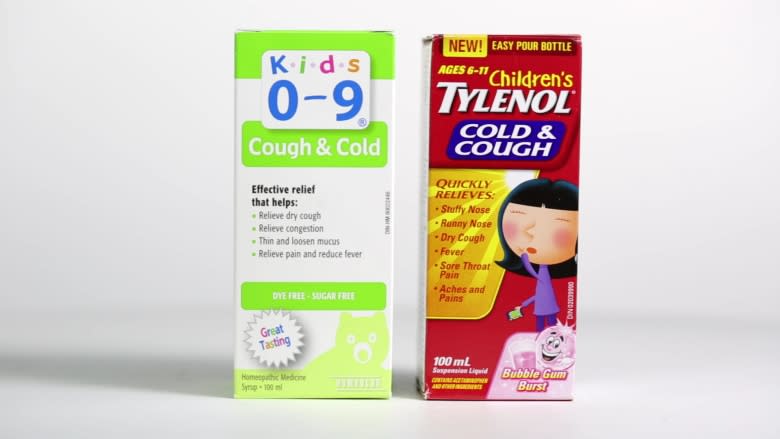
Marketplace created a children’s fever and pain remedy called Nighton, which claimed to provide "effective relief from fever, pain, and inflammation" for children and infants.
There is a growing market in natural children’s treatments. In 2008, Health Canada advised that over-the-counter cough and cold pharmaceutical products should not be given to children under the age of six, leading to a boom in natural alternatives as parents look for options on drugstore shelves.
To get a licence, Marketplace submitted an application in May, 2014, to Health Canada, and included photocopied pages from A Dictionary of Practical Materia Medica by Dr. John Henry Clarke, a 1902 homeopathic reference book of ingredients, as evidence for its effectiveness.
In October, Health Canada approved the application for Nighton. (The product remains licensed, but was never manufactured or offered for sale.)
Stanbrook, who is also an assistant professor of medicine and health policy at the University of Toronto, says that Health Canada’s licensing of Nighton "really makes a joke of the regulatory process."
It’s particularly concerning, he says, when products don’t live up to their claims. For instance, in the case of Nighton, ineffective treatment for fever can lead to seizures in children.
Licensing products provides 'choices'
Health Canada has been regulating natural health products since 2004 under what is now called the Natural and Non-prescription Health Products Directorate, as a way to monitor the safety of products on the market.
The agency declined to be interviewed by Marketplace about Nighton and how the agency approves natural health products. By email, Health Canada responded that licensing these products is about giving consumers more choice.
"Canadians want a range of treatment choices available to them for conditions they can manage themselves," wrote Health Canada spokesperson Eric Morrissette.
"Health Canada supports these choices in its licensing decisions by applying standards of evidence appropriate to the product type.
"In the case of your application for a homeopathic medicine number, the appropriate evidence was provided for this type of product, so the application was approved."
Some natural health products do require additional clinical evidence in order to get approval, such as products that aren’t based on homeopathic or traditional use.
Approvals face criticism
In an editorial in the CMAJ in 2011, Stanbrook wrote that Health Canada licensing of natural health products "has created a loophole through which manufacturers can sell a product with implied health benefits without having to obtain the supporting scientific evidence that would be needed if it were sold as a drug."
There should be one standard of approval for products if a manufacturer wants to make a health claim on packaging, Stanbrook says, "If you want to sell a natural health product and you don't want to do the science to prove that it works, then don't claim that it works."
Existing regulations allow products "to be marketed with ... poorly documented health benefits and little or no safety data," he wrote. And many companies "seem all too willing to convey misinformation about the ostensible benefits of natural or other nondrug products."
Health Canada has faced similar criticism from public health advocates in recent months over its licensing of nosodes, which are treatments that some homeopaths sell as vaccine alternatives. There is no evidence that nosodes provide immunity against disease.
In the fall of 2014, Marketplace investigated homeopathic practitioners selling homeopathic nosodes as an alternative to conventional vaccines. The investigation found that some practitioners downplayed the dangers of disease, while saying that nosodes were 95 per cent effective against diseases including measles and whooping cough.
Health Canada has licensed nosodes but says they are not approved as "a substitute for conventional vaccines."